Preclinical and Clinical Research
Good Clinical Practices (GCP)
GCP Audit is a systematic check-up, by our expert Auditors to evaluate whether the implementation, data recording and analysis of the trial are in accordance with the trial protocol, standard operation procedure and other regulations related to drug and device clinical trials
GCP Audits are spread across Clinical Trials, Bioavailability and Bioequivalence studies for New Drug Application (NDA) and Abbreviated New Drug Application (ANDA) at various stages of drug developmental process from Phase I-IV in Healthy population and Patient Population
GCP Audits are applicable in broad range of any interventions or non-interventions of food additives, drug, biological, electronic products, medical devices for human application
GCP audit can be a full clinical trial audit over systems, quality management plan and operations conducted by our third party monitors, a full clinical trial audit over the clinical trial sites, or an audit of the CRO carrying out the clinical trial on behalf of the sponsor
GCP Audit will be conducted in line with principles of ICH GCP, CDSCO, ICMR guidance, and other international regulatory guidelines, principles to which sponsor is intended to submit their dossier

Good Laboratory Practices (GLP)
GLP Audit is a quality system that covers the organizational process and the conditions under which non-clinical laboratory studies are planned, performed, monitored, recorded, archived and reported
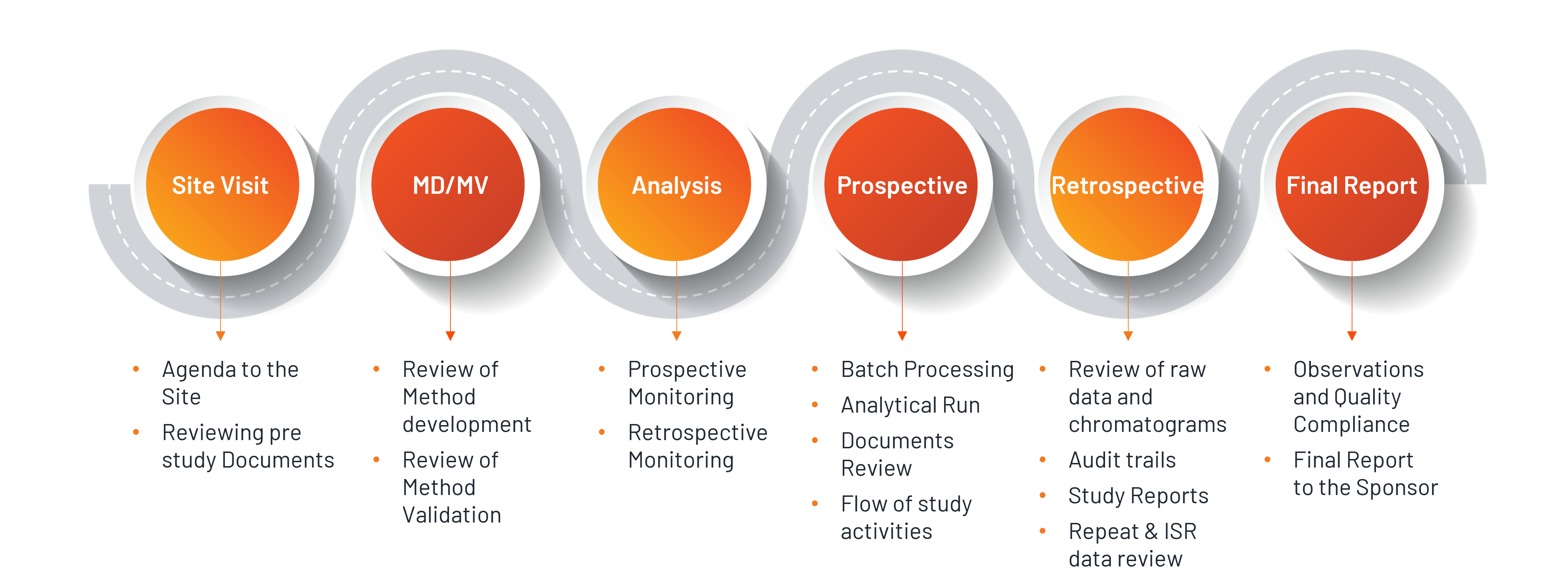
Our GLP Audits are broadening from Non-Clinical laboratory to Clinical Bioanalysis, though the, clinical bioanalysis not required to monitor under GLP program. The Bioanalysis part of Bioequivalence studies are very important in evaluating the study out comes and should be performed in accordance with the principles of GLP
We as a Third party agency, ensures in vivo or in vitro experiments in which test articles are studied under laboratory conditions to determine their safety across various stages of drug development from Investigation New Drug Application (IND), New Drug Application (NDA), 505(b)(2)
GLP Audits are applicable in broad range of any interventions or non-interventions of food additives, drug, biological, electronic products, medical devices for human application in preclinical and clinical laboratory stages
GLP Audit will be conducted in line with principles of OECD, Good Laboratory Practices Compliance Authority by the department of Science and technology Govt. of India and other international regulatory guidelines, principles to which sponsor is intended to submit their dossier